Phase II, Double-Blinded, Randomized, Placebo-Controlled Clinical Trial Investigating the Efficacy of Mebendazole in the Management of Symptomatic COVID-19 Patients
Mohamed El-Tanani, Khaled Abdul-Aziz Ahmed, Ashok K Shakya, Wesam G Ammari, Abdel-Elah Al-Shudifat
Pharmaceuticals, doi:10.3390/ph16060799
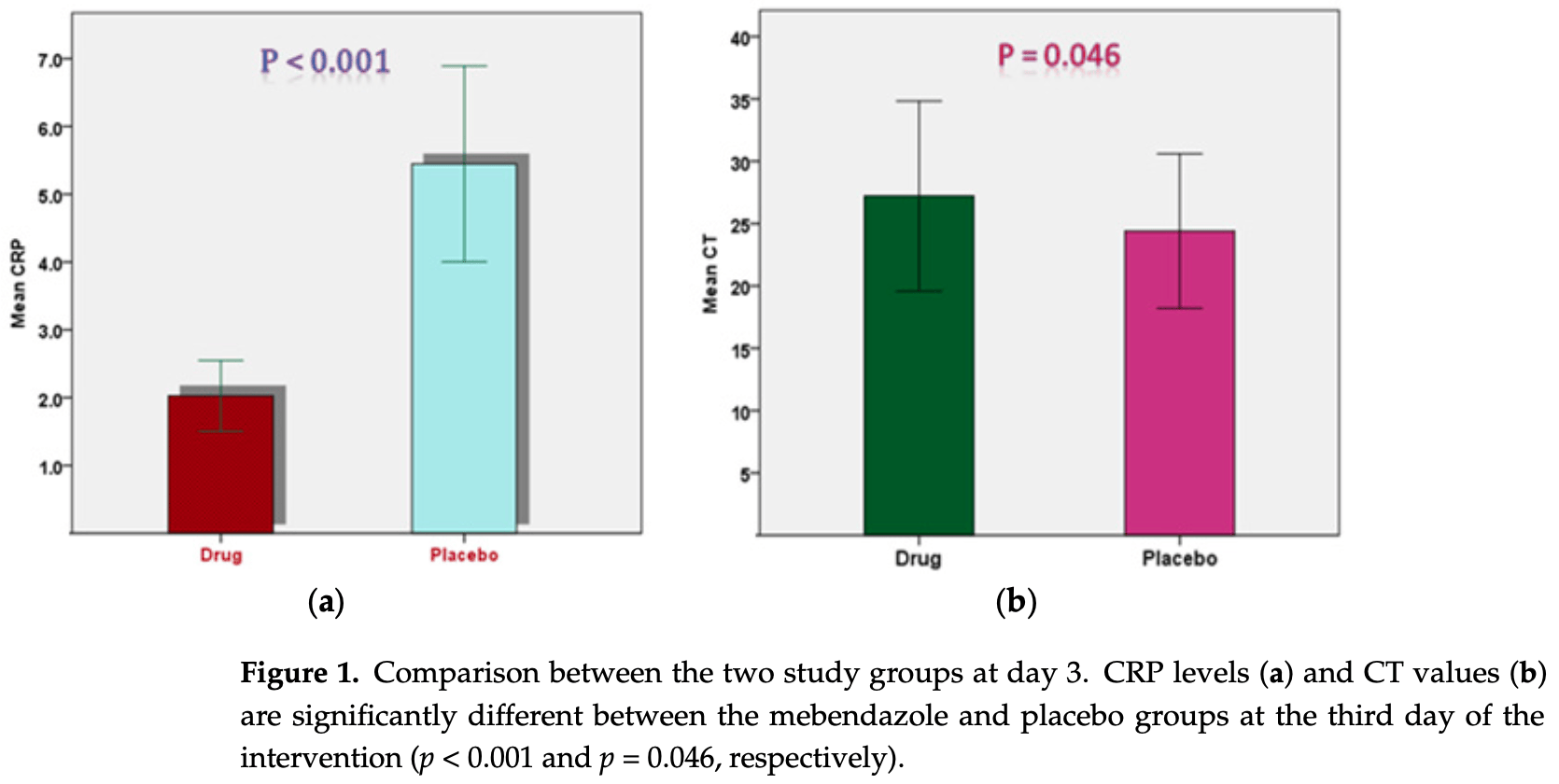
The outbreak of the COVID-19 pandemic has spread throughout the world, affecting almost all nations and territories. The current double-blind, randomized, placebo-controlled, phase II clinical trial sought to evaluate the clinical efficacy and safety of mebendazole as an adjuvant therapy for outpatients with COVID-19. The patients were recruited and divided into two groups: a Mebendazole-treated group and placebo group. The mebendazole and placebo groups were matched for age, sex, and complete blood count (CBC) with differential and liver and kidney function tests at baseline. On the third day, the C-reactive protein (CRP) levels were lower (2.03 ± 1.45 vs. 5.45 ± 3.95, p < 0.001) and the cycle threshold (CT) levels were higher (27.21 ± 3.81 vs. 24.40 ± 3.09, p = 0.046) significantly in the mebendazole group than in the placebo group on the third day. Furthermore, CRP decreased and CT dramatically increased on day three compared to the baseline day in the mebendazole group (p < 0.001 and p = 0.008, respectively). There was a significant inverse correlation between lymphocytes and CT levels in the mebendazole group (r = -0.491, p = 0.039) but not in the placebo group (r = 0.051, p = 0.888). Mebendazole therapy increased innate immunity and returned inflammation to normal levels in COVID-19 outpatients faster than it did in the placebo group in this clinical trial. Our findings add to the growing body of research on the clinical and microbiological benefits of repurposing antiparasitic therapy, specifically mebendazole, for SARS-CoV-2 infection and other viral infections.
Conflicts of Interest: The authors declare no conflict of interest.
References
Ahmed, Farag, Boys, Wang, Menendez-Montes et al., FDA approved drugs with antiviral activity against SARS-CoV-2: From structure-based repurposing to host-specific mechanisms, Biomed. Pharmacother,
doi:10.1016/j.biopha.2023.114614Al-Balas, Al-Balas, Alqassieh, Al-Balas, Al-Balas et al., Clinical features of COVID-19 patients in Jordan: A study of 508 patients, Open Respir. Med. J. 2021,
doi:10.2174/1874306402115010028Alamer, Alrashed, Alfaifi, Alosaimi, Alhassar et al., Effectiveness and safety of favipiravir compared to supportive care in moderately to critically ill COVID-19 patients: A retrospective study with propensity score matching sensitivity analysis, Curr. Med. Res. Opin,
doi:10.1080/03007995.2021.1920900Andersson, Selvin, Blom, Rubin, Berglund et al., Mebendazole is unique among tubulin-active drugs in activating the MEK-ERK pathway, Sci. Rep,
doi:10.1038/s41598-020-68986-0Blom, Rubin, Berglund, Jarvius, Lenhammar et al., Mebendazole-induced M1 polarisation of THP-1 macrophages may involve DYRK1B inhibition, BMC Res. Notes,
doi:10.1186/s13104-019-4273-5Bosaeed, Alharbi, Hussein, Abalkhail, Sultana et al., Multicentre randomised double-blinded placebo-controlled trial of favipiravir in adults with mild COVID-19, BMJ Open,
doi:10.1136/bmjopen-2020-047495Bosaeed, Alharbi, Mahmoud, Alrehily, Bahlaq et al., Efficacy of favipiravir in adults with mild COVID-19: A randomized, double-blind, multicentre, placebo-controlled clinical trial, Clin. Microbiol. Infect,
doi:10.1016/j.cmi.2021.12.026Chai, Jung, Hong, Albendazole and mebendazole as anti-parasitic and anti-cancer agents: An update, Korean J. Parasitol,
doi:10.3347/kjp.2021.59.3.189Chen, Hu, Hood, Zhang, Zhang et al., A novel combination of vitamin C, curcumin and glycyrrhizic acid potentially regulates immune and inflammatory response associated with coronavirus infections: A perspective from system biology analysis, Nutrients,
doi:10.3390/nu12041193Covid-19, Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines, National Institutes of Health: Bethesda
De Witt, Gamble, Hanson, Markowitz, Powell et al., Repurposing mebendazole as a replacement for vincristine for the treatment of brain tumors, Mol. Med
Domenico, Vanelli, WHO Declares COVID-19 a Pandemic, Acta Biomed
Dorward, Yu, Hayward, Saville, Gbinigie et al., Colchicine for COVID-19 in the community (PRINCIPLE): A randomised, controlled, adaptive platform trial, Br. J. Gen. Pract,
doi:10.3399/BJGP.2022.0083Elayapillai, Ramraj, Benbrook, Bieniasz, Wang et al., Potential and mechanism of mebendazole for treatment and maintenance of ovarian cancer, Gynecol. Oncol,
doi:10.1016/j.ygyno.2020.10.010Farag, Wang, Boys, Eitson, Ohlson et al., Identification of Atovaquone, Ouabain and Mebendazole as FDA Approved Drugs Targeting SARS-CoV-2 (Version 4), ChemRxiv,
doi:10.26434/chemrxiv.12003930.v4Ferrari, Seveso, Sabetta, Ceriotti, Carobene et al., Role of time-normalized laboratory findings in predicting COVID-19 outcome, Diagnosis,
doi:10.1515/dx-2020-0095Galal, Ahmed, Shao, Xing, Ali et al., The Use of Mebendazole in COVID-19 Patients: An Observational Retrospective Single Center Study, Adv. Virol,
doi:10.1155/2022/3014686Guerini, Triggiani, Maddalo, Bonù, Frassine et al., Mebendazole as a candidate for drug repurposing in oncology: An extensive review of current literature, Cancers,
doi:10.3390/cancers11091284Hajjo, Tropsha, A systems biology workflow for drug and vaccine repurposing: Identifying small-molecule BCG mimics to reduce or prevent COVID-19 mortality, Pharm. Res,
doi:10.1007/s11095-020-02930-9Hegazy, El-Azab, Zakaria, Mostafa, El-Ghoneimy et al., from an anti-parasitic drug to a promising candidate for drug repurposing in colorectal cancer, Life Sci,
doi:10.1016/j.lfs.2022.120536Hemphill, Müller, Alveolar and cystic echinococcosis: Towards novel chemotherapeutical treatment options, J. Helminthol,
doi:10.1017/S0022149X0928936XKaruppagounder, Zhai, Chen, He, Ratan, The interferon response as a common final pathway for many preconditioning stimuli: Unexpected crosstalk between hypoxic adaptation and antiviral defense, Cond. Med
Khader, Al Nsour, Excess mortality during the COVID-19 pandemic in Jordan: Secondary data analysis,
doi:10.2196/32559Kudo, Kubota, Gotoh, Ishida, Ikadai et al., Efficacy of thiabendazole, mebendazole, levamisole and ivermectin against gullet worm, Gongylonema pulchrum: In vitro and in vivo studies, Vet. Parasitol,
doi:10.1016/j.vetpar.2007.10.005Law, Akers, Tasnina, Santina, Deutsch et al., Interpretable network propagation with application to expanding the repertoire of human proteins that interact with SARS-CoV-2,
doi:10.1093/gigascience/giab082Li, Thomas, Deutzmann, Majeti, Felsher et al., Mebendazole for differentiation therapy of acute myeloid leukemia identified by a lineage maturation index, Sci. Rep,
doi:10.1038/s41598-019-53290-3Murer, Volle, Andriasyan, Petkidis, Gomez-Gonzalez et al., Identification of broad anti-coronavirus chemical agents for repurposing against SARS-CoV-2 and variants of concern, Curr. Res. Virol. Sci,
doi:10.1016/j.crviro.2022.100019Panahi, Dadkhah, Talei, Gharari, Asghariazar et al., Can anti-parasitic drugs help control COVID-19? Future Virol,
doi:10.2217/fvl-2021-0160Patel, Zhang, Fullerton, Boelen, Rongvaux et al., The fate and lifespan of human monocyte subsets in steady state and systemic inflammation, J. Exp. Med,
doi:10.1084/jem.20170355Puente, Lago, Subirats, Sanz-Esteban, Arsuaga et al., Imported Mansonella perstans infection in Spain, Infect. Dis. Poverty,
doi:10.1186/s40249-020-00729-9Reis, Silva, Silva, Thabane, Milagres et al., Effect of early treatment with ivermectin among patients with COVID-19, N. Engl. J. Med,
doi:10.1056/NEJMoa2115869Reis, Silva, Silva, Thabane, Singh et al., Effect of early treatment with hydroxychloroquine or lopinavir and ritonavir on risk of hospitalization among patients with COVID-19: The TOGETHER randomized clinical trial, JAMA Netw,
doi:10.1001/jamanetworkopen.2021.6468Rushworth, Hewit, Munnings-Tomes, Somani, James et al., Repurposing screen identifies mebendazole as a clinical candidate to synergise with docetaxel for prostate cancer treatment, Br. J. Cancer,
doi:10.1038/s41416-019-0681-5Seitz, Schwerk, Arnold, Hepatocellular drug reaction caused by mebendazole therapy in cystic echinococcosis, Z. Gastroenterol
Sun, Wang, Cai, Hu, Liao et al., Cytokine storm intervention in the early stages of COVID-19 pneumonia, Cytokine Growth Factor. Rev,
doi:10.1016/j.cytogfr.2020.04.002Surapat, Kobpetchyok, Kiertiburanakul, Arnuntasupakul, Use of Favipiravir for the Treatment of Coronavirus Disease 2019 in the Setting of Hospitel, Int. J. Clin. Pract,
doi:10.1155/2022/3098527Tolomeo, Colomba, Meli, Cascio, Hepatotoxicity caused by mebendazole in a patient with Gilbert's syndrome, J. Clin. Pharm. Ther,
doi:10.1111/jcpt.13033Tonelli, Simone, Tasso, Novelli, Boido et al., Antiviral activity of benzimidazole derivatives. II. Antiviral activity of 2-phenylbenzimidazole derivatives, Bioorg. Med. Chem,
doi:10.1016/j.bmc.2010.02.037Turner, Shamseer, Altman, Weeks, Peters et al., Consolidated standards of reporting trials (CONSORT) and the completeness of reporting of randomised controlled trials (RCTs) published in medical journals, Cochrane Database Syst. Rev,
doi:10.1002/14651858.MR000030.pub2Udwadia, Singh, Barkate, Patil, Rangwala et al., Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial, Int. J. Infect. Dis,
doi:10.1016/j.ijid.2020.11.142Villoteau, Asfar, Otekpo, Loison, Gautier et al., GERIA-COVID study group, Elevated C-reactive protein in early COVID-19 predicts worse survival among hospitalized geriatric patients, PLoS ONE,
doi:10.1371/journal.pone.0256931Wang, Guo, Gao, Pu, Li et al., Repurposable drugs for SARS-CoV-2 and influenza sepsis with scRNA-seq data targeting post-transcription modifications. Precis, Clin. Med,
doi:10.1093/pcmedi/pbab022Worldometer, Cwg, Coronavirus Worldwide Graphs. Available online
Yamakawa, Yamamoto, Terayama, Hashimoto, Ishihara et al., Japanese Rapid/Living recommendations on drug management for COVID-19,
doi:10.1002/ams2.664Yele, Sanapalli, Mohammed, Imidazoles and benzimidazoles as putative inhibitors of SARS-CoV-2 B. 1.1. 7 (Alpha) and P. 1 (Gamma) variant spike glycoproteins: A computational approach, Chem. Zvesti
Zhang, Bochkur, Yazal, Dong, Nguyen et al., Mebendazole potentiates radiation therapy in triple-negative breast cancer, Int. J. Radiat. Oncol. Biol. Phys,
doi:10.1016/j.ijrobp.2018.08.046DOI record:
{
"DOI": "10.3390/ph16060799",
"ISSN": [
"1424-8247"
],
"URL": "http://dx.doi.org/10.3390/ph16060799",
"abstract": "<jats:p>The outbreak of the COVID-19 pandemic has spread throughout the world, affecting almost all nations and territories. The current double-blind, randomized, placebo-controlled, phase II clinical trial sought to evaluate the clinical efficacy and safety of mebendazole as an adjuvant therapy for outpatients with COVID-19. The patients were recruited and divided into two groups: a Mebendazole-treated group and placebo group. The mebendazole and placebo groups were matched for age, sex, and complete blood count (CBC) with differential and liver and kidney function tests at baseline. On the third day, the C-reactive protein (CRP) levels were lower (2.03 ± 1.45 vs. 5.45 ± 3.95, p < 0.001) and the cycle threshold (CT) levels were higher (27.21 ± 3.81 vs. 24.40 ± 3.09, p = 0.046) significantly in the mebendazole group than in the placebo group on the third day. Furthermore, CRP decreased and CT dramatically increased on day three compared to the baseline day in the mebendazole group (p < 0.001 and p = 0.008, respectively). There was a significant inverse correlation between lymphocytes and CT levels in the mebendazole group (r = −0.491, p = 0.039) but not in the placebo group (r = 0.051, p = 0.888). Mebendazole therapy increased innate immunity and returned inflammation to normal levels in COVID-19 outpatients faster than it did in the placebo group in this clinical trial. Our findings add to the growing body of research on the clinical and microbiological benefits of repurposing antiparasitic therapy, specifically mebendazole, for SARS-CoV-2 infection and other viral infections.</jats:p>",
"alternative-id": [
"ph16060799"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-4735-5445",
"affiliation": [
{
"name": "Pharmacological and Diagnostic Research Centre (PDRC), Al-Ahliyya Amman University, Amman 19328, Jordan"
},
{
"name": "Institute of Cancer Therapeutics, Faculty of Life Sciences, University of Bradford, Bradford BD7 1DP, UK"
}
],
"authenticated-orcid": false,
"family": "El-Tanani",
"given": "Mohamed",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Pharmacological and Diagnostic Research Centre (PDRC), Al-Ahliyya Amman University, Amman 19328, Jordan"
},
{
"name": "Department of Medical Laboratory Sciences, Faculty of Allied Medical Sciences, Al-Ahliyya Amman University, Amman 19328, Jordan"
}
],
"family": "Ahmed",
"given": "Khaled Abdul-Aziz",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-2387-6940",
"affiliation": [
{
"name": "Pharmacological and Diagnostic Research Centre (PDRC), Al-Ahliyya Amman University, Amman 19328, Jordan"
}
],
"authenticated-orcid": false,
"family": "Shakya",
"given": "Ashok K.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-0547-8428",
"affiliation": [
{
"name": "Pharmacological and Diagnostic Research Centre (PDRC), Al-Ahliyya Amman University, Amman 19328, Jordan"
}
],
"authenticated-orcid": false,
"family": "Ammari",
"given": "Wesam G.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Internal and Family Medicine, Faculty of Medicine, The Hashemite University, Zarqa 13133, Jordan"
}
],
"family": "Al-Shudifat",
"given": "Abdel-Elah",
"sequence": "additional"
}
],
"container-title": "Pharmaceuticals",
"container-title-short": "Pharmaceuticals",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2023,
5,
29
]
],
"date-time": "2023-05-29T11:54:16Z",
"timestamp": 1685361256000
},
"deposited": {
"date-parts": [
[
2023,
5,
29
]
],
"date-time": "2023-05-29T13:40:26Z",
"timestamp": 1685367626000
},
"funder": [
{
"DOI": "10.13039/501100016299",
"award": [
"2021/12/19"
],
"doi-asserted-by": "publisher",
"name": "Al-Ahliyya Amman University, Jordan"
}
],
"indexed": {
"date-parts": [
[
2023,
5,
30
]
],
"date-time": "2023-05-30T04:28:04Z",
"timestamp": 1685420884065
},
"is-referenced-by-count": 0,
"issue": "6",
"issued": {
"date-parts": [
[
2023,
5,
29
]
]
},
"journal-issue": {
"issue": "6",
"published-online": {
"date-parts": [
[
2023,
6
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2023,
5,
29
]
],
"date-time": "2023-05-29T00:00:00Z",
"timestamp": 1685318400000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/1424-8247/16/6/799/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "799",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2023,
5,
29
]
]
},
"published-online": {
"date-parts": [
[
2023,
5,
29
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"DOI": "10.1016/j.scitotenv.2020.138882",
"article-title": "COVID-19 outbreak: Migration, effects on society, global environment and prevention",
"author": "Chakraborty",
"doi-asserted-by": "crossref",
"first-page": "138882",
"journal-title": "Sci. Total Environ.",
"key": "ref_1",
"volume": "728",
"year": "2020"
},
{
"article-title": "WHO Declares COVID-19 a Pandemic",
"author": "Domenico",
"first-page": "157",
"journal-title": "Acta Biomed.",
"key": "ref_2",
"volume": "91",
"year": "2020"
},
{
"key": "ref_3",
"unstructured": "Worldometer (2022, May 10). COVID-19 Coronavirus Pandemic. Available online: https://www.worldometers.info/coronavirus/."
},
{
"key": "ref_4",
"unstructured": "Worldometer (2022, May 25). CWG.Coronavirus Worldwide Graphs. Available online: https://www.worldometers.info/coronavirus/worldwide-graphs/#total-deaths."
},
{
"key": "ref_5",
"unstructured": "World Health Organization (2022, May 11). Jordan: WHO Coronavirus Disease (COVID-19) Dashboard with Vaccination Data. Available online: https://covid19.who.int/region/emro/country/jo."
},
{
"DOI": "10.2196/32559",
"article-title": "Excess mortality during the COVID-19 pandemic in Jordan: Secondary data analysis",
"author": "Khader",
"doi-asserted-by": "crossref",
"first-page": "e32559",
"journal-title": "JMIR Public Health Surveill.",
"key": "ref_6",
"volume": "7",
"year": "2021"
},
{
"DOI": "10.2174/1874306402115010028",
"article-title": "Clinical features of COVID-19 patients in Jordan: A study of 508 patients",
"author": "Alqassieh",
"doi-asserted-by": "crossref",
"first-page": "28",
"journal-title": "Open Respir. Med. J.",
"key": "ref_7",
"volume": "15",
"year": "2021"
},
{
"DOI": "10.1016/S0140-6736(20)30183-5",
"article-title": "Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China",
"author": "Huang",
"doi-asserted-by": "crossref",
"first-page": "497",
"journal-title": "Lancet",
"key": "ref_8",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1016/S2213-2600(22)00213-2",
"article-title": "Early treatment to prevent progression of SARS-CoV-2 infection",
"author": "Cohen",
"doi-asserted-by": "crossref",
"first-page": "P930",
"journal-title": "Lancet Respir. Med.",
"key": "ref_9",
"volume": "10",
"year": "2022"
},
{
"DOI": "10.1080/03007995.2021.1920900",
"article-title": "Effectiveness and safety of favipiravir compared to supportive care in moderately to critically ill COVID-19 patients: A retrospective study with propensity score matching sensitivity analysis",
"author": "Alamer",
"doi-asserted-by": "crossref",
"first-page": "1085",
"journal-title": "Curr. Med. Res. Opin.",
"key": "ref_10",
"volume": "37",
"year": "2021"
},
{
"DOI": "10.1016/j.ijid.2020.11.142",
"article-title": "Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial",
"author": "Udwadia",
"doi-asserted-by": "crossref",
"first-page": "62",
"journal-title": "Int. J. Infect. Dis.",
"key": "ref_11",
"volume": "103",
"year": "2021"
},
{
"key": "ref_12",
"unstructured": "(2021, November 07). Ministry of Health Saudi Arabia, Available online: https://www.moh.gov.sa/en/Pages/Default.aspx."
},
{
"article-title": "Japanese Rapid/Living recommendations on drug management for COVID-19",
"author": "Yamakawa",
"first-page": "e789",
"journal-title": "Acute Med. Surg.",
"key": "ref_13",
"volume": "9",
"year": "2021"
},
{
"DOI": "10.1016/j.cmi.2021.12.026",
"article-title": "Efficacy of favipiravir in adults with mild COVID-19: A randomized, double-blind, multicentre, placebo-controlled clinical trial",
"author": "Bosaeed",
"doi-asserted-by": "crossref",
"first-page": "602",
"journal-title": "Clin. Microbiol. Infect.",
"key": "ref_14",
"volume": "28",
"year": "2022"
},
{
"DOI": "10.1016/j.bjid.2020.06.002",
"article-title": "Ivermectin: Potential candidate for the treatment of COVID-19",
"author": "Gupta",
"doi-asserted-by": "crossref",
"first-page": "369",
"journal-title": "Braz. J. Infect. Dis.",
"key": "ref_15",
"volume": "24",
"year": "2020"
},
{
"DOI": "10.1016/j.cytogfr.2020.04.002",
"article-title": "Cytokine storm intervention in the early stages of COVID-19 pneumonia",
"author": "Sun",
"doi-asserted-by": "crossref",
"first-page": "38",
"journal-title": "Cytokine Growth Factor. Rev.",
"key": "ref_16",
"volume": "53",
"year": "2020"
},
{
"DOI": "10.3390/nu12041193",
"doi-asserted-by": "crossref",
"key": "ref_17",
"unstructured": "Chen, L., Hu, C., Hood, M., Zhang, X., Zhang, L., Kan, J., and Du, J. (2020). A novel combination of vitamin C, curcumin and glycyrrhizic acid potentially regulates immune and inflammatory response associated with coronavirus infections: A perspective from system biology analysis. Nutrients, 12."
},
{
"DOI": "10.1155/2022/3014686",
"article-title": "The Use of Mebendazole in COVID-19 Patients: An Observational Retrospective Single Center Study",
"author": "Galal",
"doi-asserted-by": "crossref",
"first-page": "3014686",
"journal-title": "Adv. Virol.",
"key": "ref_18",
"volume": "2022",
"year": "2022"
},
{
"DOI": "10.7326/0003-4819-91-4-582",
"article-title": "Drugs Five Years Later: Mebendazole",
"author": "Keystone",
"doi-asserted-by": "crossref",
"first-page": "582",
"journal-title": "Ann. Intern. Med.",
"key": "ref_19",
"volume": "91",
"year": "1979"
},
{
"DOI": "10.3390/cancers11091284",
"doi-asserted-by": "crossref",
"key": "ref_20",
"unstructured": "Guerini, A.E., Triggiani, L., Maddalo, M., Bonù, M.L., Frassine, F., Baiguini, A., Alghisi, A., Tomasini, D., Borghetti, P., and Pasinetti, N. (2019). Mebendazole as a candidate for drug repurposing in oncology: An extensive review of current literature. Cancers, 11."
},
{
"DOI": "10.1016/j.ygyno.2020.10.010",
"article-title": "Potential and mechanism of mebendazole for treatment and maintenance of ovarian cancer",
"author": "Elayapillai",
"doi-asserted-by": "crossref",
"first-page": "302",
"journal-title": "Gynecol. Oncol.",
"key": "ref_21",
"volume": "160",
"year": "2021"
},
{
"DOI": "10.1038/s41416-019-0681-5",
"article-title": "Repurposing screen identifies mebendazole as a clinical candidate to synergise with docetaxel for prostate cancer treatment",
"author": "Rushworth",
"doi-asserted-by": "crossref",
"first-page": "517",
"journal-title": "Br. J. Cancer",
"key": "ref_22",
"volume": "122",
"year": "2020"
},
{
"DOI": "10.2119/molmed.2017.00011",
"article-title": "Repurposing mebendazole as a replacement for vincristine for the treatment of brain tumors",
"author": "Gamble",
"doi-asserted-by": "crossref",
"first-page": "50",
"journal-title": "Mol. Med.",
"key": "ref_23",
"volume": "23",
"year": "2017"
},
{
"DOI": "10.1038/s41598-019-53290-3",
"article-title": "Mebendazole for differentiation therapy of acute myeloid leukemia identified by a lineage maturation index",
"author": "Li",
"doi-asserted-by": "crossref",
"first-page": "16775",
"journal-title": "Sci. Rep.",
"key": "ref_24",
"volume": "9",
"year": "2019"
},
{
"DOI": "10.1016/j.ijrobp.2018.08.046",
"article-title": "Mebendazole potentiates radiation therapy in triple-negative breast cancer",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "195",
"journal-title": "Int. J. Radiat. Oncol. Biol. Phys.",
"key": "ref_25",
"volume": "103",
"year": "2019"
},
{
"DOI": "10.1016/j.lfs.2022.120536",
"article-title": "Mebendazole; from an anti-parasitic drug to a promising candidate for drug repurposing in colorectal cancer",
"author": "Hegazy",
"doi-asserted-by": "crossref",
"first-page": "120536",
"journal-title": "Life Sci.",
"key": "ref_26",
"volume": "299",
"year": "2022"
},
{
"DOI": "10.1016/j.vetpar.2007.10.005",
"article-title": "Efficacy of thiabendazole, mebendazole, levamisole and ivermectin against gullet worm, Gongylonema pulchrum: In vitro and in vivo studies",
"author": "Kudo",
"doi-asserted-by": "crossref",
"first-page": "46",
"journal-title": "Vet. Parasitol.",
"key": "ref_27",
"volume": "151",
"year": "2008"
},
{
"DOI": "10.1017/S0022149X0928936X",
"article-title": "Alveolar and cystic echinococcosis: Towards novel chemotherapeutical treatment options",
"author": "Hemphill",
"doi-asserted-by": "crossref",
"first-page": "99",
"journal-title": "J. Helminthol.",
"key": "ref_28",
"volume": "83",
"year": "2009"
},
{
"DOI": "10.1186/s13104-019-4273-5",
"doi-asserted-by": "crossref",
"key": "ref_29",
"unstructured": "Blom, K., Rubin, J., Berglund, M., Jarvius, M., Lenhammar, L., Parrow, V., Andersson, C., Loskog, A., Fryknäs, M., and Nygren, P. (2019). Mebendazole-induced M1 polarisation of THP-1 macrophages may involve DYRK1B inhibition. BMC Res. Notes, 12."
},
{
"DOI": "10.1038/s41598-020-68986-0",
"article-title": "Mebendazole is unique among tubulin-active drugs in activating the MEK–ERK pathway",
"author": "Andersson",
"doi-asserted-by": "crossref",
"first-page": "13124",
"journal-title": "Sci. Rep.",
"key": "ref_30",
"volume": "10",
"year": "2020"
},
{
"article-title": "The interferon response as a common final pathway for many preconditioning stimuli: Unexpected crosstalk between hypoxic adaptation and antiviral defense",
"author": "Karuppagounder",
"first-page": "143",
"journal-title": "Cond. Med.",
"key": "ref_31",
"volume": "1",
"year": "2018"
},
{
"DOI": "10.1093/gigascience/giab082",
"article-title": "Interpretable network propagation with application to expanding the repertoire of human proteins that interact with SARS-CoV-2",
"author": "Law",
"doi-asserted-by": "crossref",
"first-page": "giab082",
"journal-title": "GigaScience",
"key": "ref_32",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.1007/s11095-020-02930-9",
"article-title": "A systems biology workflow for drug and vaccine repurposing: Identifying small-molecule BCG mimics to reduce or prevent COVID-19 mortality",
"author": "Hajjo",
"doi-asserted-by": "crossref",
"first-page": "212",
"journal-title": "Pharm. Res.",
"key": "ref_33",
"volume": "37",
"year": "2020"
},
{
"key": "ref_34",
"unstructured": "National Institute of Diabetes and Digestive and Kidney Diseases (2012). LiverTox: Clinical and Research Information on Drug-Induced Liver Injury."
},
{
"DOI": "10.1080/00034983.1982.11687523",
"article-title": "Mebendazole and alveolar hydatid disease",
"author": "Wilson",
"doi-asserted-by": "crossref",
"first-page": "165",
"journal-title": "Ann. Trop. Med. Parasitol.",
"key": "ref_35",
"volume": "76",
"year": "1982"
},
{
"DOI": "10.1111/jcpt.13033",
"article-title": "Hepatotoxicity caused by mebendazole in a patient with Gilbert’s syndrome",
"author": "Tolomeo",
"doi-asserted-by": "crossref",
"first-page": "985",
"journal-title": "J. Clin. Pharm. Ther.",
"key": "ref_36",
"volume": "44",
"year": "2019"
},
{
"DOI": "10.3347/kjp.2021.59.3.189",
"article-title": "Albendazole and mebendazole as anti-parasitic and anti-cancer agents: An update",
"author": "Chai",
"doi-asserted-by": "crossref",
"first-page": "189",
"journal-title": "Korean J. Parasitol.",
"key": "ref_37",
"volume": "59",
"year": "2021"
},
{
"article-title": "Hepatocellular drug reaction caused by mebendazole therapy in cystic echinococcosis",
"author": "Seitz",
"first-page": "324",
"journal-title": "Z. Gastroenterol.",
"key": "ref_38",
"volume": "21",
"year": "1983"
},
{
"DOI": "10.1111/j.1445-2197.1985.tb00935.x",
"article-title": "Hydatid disease: The alveolar variety in Australia. A case report with comment on the toxicity of mebendazole",
"author": "Braithwaite",
"doi-asserted-by": "crossref",
"first-page": "519",
"journal-title": "Aust. N. Z. J. Surg.",
"key": "ref_39",
"volume": "55",
"year": "1985"
},
{
"DOI": "10.1186/s40249-020-00729-9",
"article-title": "Imported Mansonella perstans infection in Spain",
"author": "Puente",
"doi-asserted-by": "crossref",
"first-page": "105",
"journal-title": "Infect. Dis. Poverty",
"key": "ref_40",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1001/jamanetworkopen.2021.6468",
"article-title": "Effect of early treatment with hydroxychloroquine or lopinavir and ritonavir on risk of hospitalization among patients with COVID-19: The TOGETHER randomized clinical trial",
"author": "Reis",
"doi-asserted-by": "crossref",
"first-page": "e216468",
"journal-title": "JAMA Netw. Open",
"key": "ref_41",
"volume": "4",
"year": "2021"
},
{
"key": "ref_42",
"unstructured": "COVID-19 Treatment Guidelines Panel (2022). Coronavirus Disease 2019 (COVID-19) Treatment Guidelines."
},
{
"DOI": "10.1183/13993003.02002-2021",
"article-title": "Lessons learnt from hydroxychloroquine/azithromycin in treatment of COVID-19",
"author": "Xu",
"doi-asserted-by": "crossref",
"first-page": "2102002",
"journal-title": "Eur. Respir. J.",
"key": "ref_43",
"volume": "59",
"year": "2022"
},
{
"DOI": "10.1056/NEJMoa2115869",
"article-title": "Effect of early treatment with ivermectin among patients with COVID-19",
"author": "Reis",
"doi-asserted-by": "crossref",
"first-page": "1721",
"journal-title": "N. Engl. J. Med.",
"key": "ref_44",
"volume": "386",
"year": "2022"
},
{
"DOI": "10.3399/BJGP.2022.0083",
"article-title": "Colchicine for COVID-19 in the community (PRINCIPLE): A randomised, controlled, adaptive platform trial",
"author": "Dorward",
"doi-asserted-by": "crossref",
"first-page": "e446",
"journal-title": "Br. J. Gen. Pract.",
"key": "ref_45",
"volume": "72",
"year": "2022"
},
{
"DOI": "10.1155/2022/3098527",
"article-title": "Use of Favipiravir for the Treatment of Coronavirus Disease 2019 in the Setting of Hospitel",
"author": "Surapat",
"doi-asserted-by": "crossref",
"first-page": "3098527",
"journal-title": "Int. J. Clin. Pract.",
"key": "ref_46",
"volume": "2022",
"year": "2022"
},
{
"DOI": "10.1136/bmjopen-2020-047495",
"article-title": "Multicentre randomised double-blinded placebo-controlled trial of favipiravir in adults with mild COVID-19",
"author": "Bosaeed",
"doi-asserted-by": "crossref",
"first-page": "e047495",
"journal-title": "BMJ Open",
"key": "ref_47",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.26434/chemrxiv.12003930",
"doi-asserted-by": "crossref",
"key": "ref_48",
"unstructured": "Farag, A., Wang, P., Boys, I.N., Eitson, J.L., Ohlson, M.B., Fan, W., McDougal, M.B., Ahmed, M., Schoggins, J.W., and Sadek, H. (2020). Identification of Atovaquone, Ouabain and Mebendazole as FDA Approved Drugs Targeting SARS-CoV-2 (Version 4). ChemRxiv."
},
{
"DOI": "10.1016/j.crviro.2022.100019",
"article-title": "Identification of broad anti-coronavirus chemical agents for repurposing against SARS-CoV-2 and variants of concern",
"author": "Murer",
"doi-asserted-by": "crossref",
"first-page": "100019",
"journal-title": "Curr. Res. Virol. Sci.",
"key": "ref_49",
"volume": "3",
"year": "2022"
},
{
"article-title": "Imidazoles and benzimidazoles as putative inhibitors of SARS-CoV-2 B. 1.1. 7 (Alpha) and P. 1 (Gamma) variant spike glycoproteins: A computational approach",
"author": "Yele",
"first-page": "1107",
"journal-title": "Chem. Zvesti.",
"key": "ref_50",
"volume": "76",
"year": "2022"
},
{
"DOI": "10.1093/pcmedi/pbab022",
"article-title": "Repurposable drugs for SARS-CoV-2 and influenza sepsis with scRNA-seq data targeting post-transcription modifications",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "215",
"journal-title": "Precis. Clin. Med.",
"key": "ref_51",
"volume": "4",
"year": "2021"
},
{
"DOI": "10.1016/j.bmc.2010.02.037",
"article-title": "Antiviral activity of benzimidazole derivatives. II. Antiviral activity of 2-phenylbenzimidazole derivatives",
"author": "Tonelli",
"doi-asserted-by": "crossref",
"first-page": "2937",
"journal-title": "Bioorg. Med. Chem.",
"key": "ref_52",
"volume": "18",
"year": "2010"
},
{
"DOI": "10.1016/j.biopha.2023.114614",
"article-title": "FDA approved drugs with antiviral activity against SARS-CoV-2: From structure-based repurposing to host-specific mechanisms",
"author": "Ahmed",
"doi-asserted-by": "crossref",
"first-page": "114614",
"journal-title": "Biomed. Pharmacother.",
"key": "ref_53",
"volume": "162",
"year": "2023"
},
{
"DOI": "10.2217/fvl-2021-0160",
"article-title": "Can anti-parasitic drugs help control COVID-19?",
"author": "Panahi",
"doi-asserted-by": "crossref",
"first-page": "315",
"journal-title": "Future Virol.",
"key": "ref_54",
"volume": "17",
"year": "2022"
},
{
"DOI": "10.1084/jem.20170355",
"article-title": "The fate and lifespan of human monocyte subsets in steady state and systemic inflammation",
"author": "Patel",
"doi-asserted-by": "crossref",
"first-page": "1913",
"journal-title": "J. Exp. Med.",
"key": "ref_55",
"volume": "214",
"year": "2017"
},
{
"DOI": "10.1515/dx-2020-0095",
"article-title": "Role of time-normalized laboratory findings in predicting COVID-19 outcome",
"author": "Ferrari",
"doi-asserted-by": "crossref",
"first-page": "387",
"journal-title": "Diagnosis",
"key": "ref_56",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1371/journal.pone.0256931",
"doi-asserted-by": "crossref",
"key": "ref_57",
"unstructured": "Villoteau, A., Asfar, M., Otekpo, M., Loison, J., Gautier, J., and Annweiler, C. (2021). GERIA-COVID study group, Elevated C-reactive protein in early COVID-19 predicts worse survival among hospitalized geriatric patients. PLoS ONE, 16."
},
{
"article-title": "Consolidated standards of reporting trials (CONSORT) and the completeness of reporting of randomised controlled trials (RCTs) published in medical journals",
"author": "Turner",
"first-page": "mr000030",
"journal-title": "Cochrane Database Syst. Rev.",
"key": "ref_58",
"volume": "11",
"year": "2012"
}
],
"reference-count": 58,
"references-count": 58,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/1424-8247/16/6/799"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Drug Discovery",
"Pharmaceutical Science",
"Molecular Medicine"
],
"subtitle": [],
"title": "Phase II, Double-Blinded, Randomized, Placebo-Controlled Clinical Trial Investigating the Efficacy of Mebendazole in the Management of Symptomatic COVID-19 Patients",
"type": "journal-article",
"volume": "16"
}