Does ivermectin reduce COVID-19 mortality and progression of disease severity? – A retrospective study.
Tehseen Akhtar, Amber Hanif, Pyar Ali, Muhammad Hussain Haroon, Nimra Akram, Muhammad Umar Raza, Khalid Imran, Nimra Shakeel, Ramsha Shakeel
The Professional Medical Journal, doi:10.29309/tpmj/2022.29.09.6634
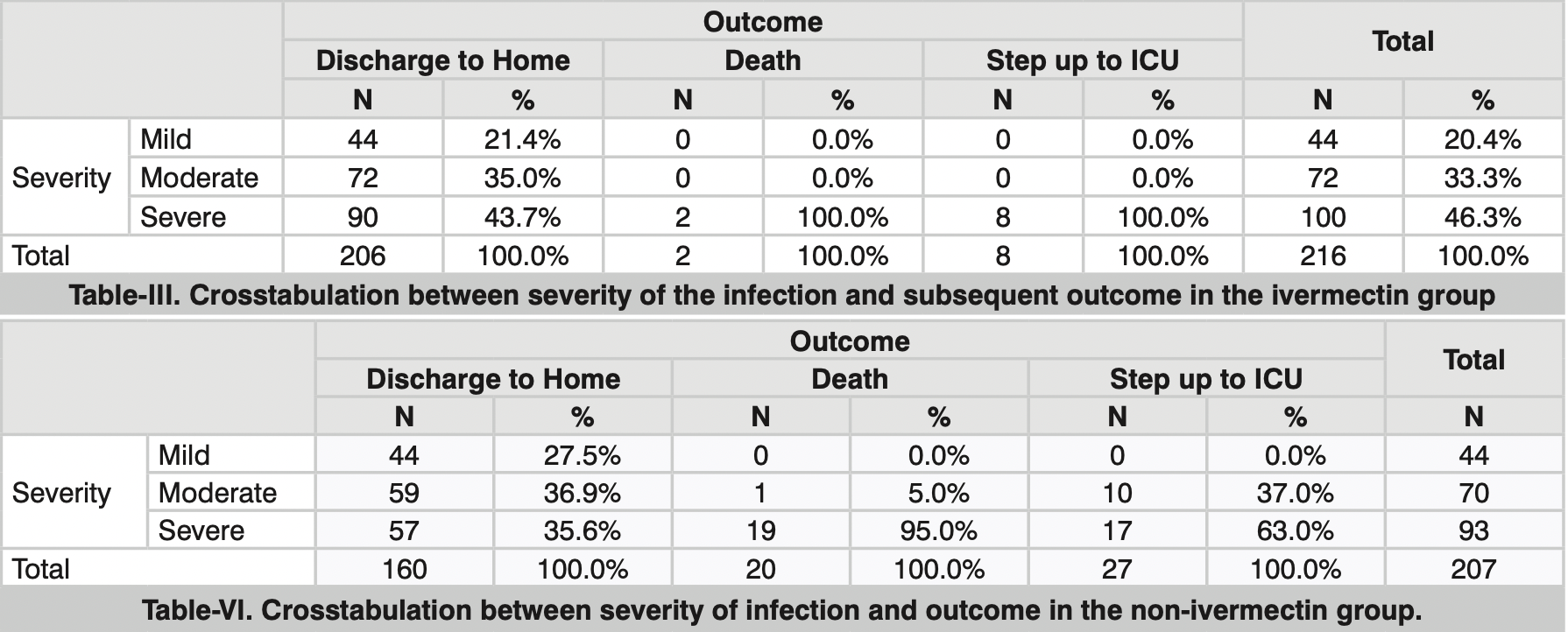
Material & Methods: Medical records of 423 patients during a selected duration of 6 months were reviewed. Patients were stratified into two groups based on whether or not they received a 6-day course of ivermectin in addition to the standard treatment for COVID-19. Primary outcome measures were rate of mortality, days from the start of treatment to negative SARS-CoV-2 PCR, and rate of step-up to the intensive care unit. Results: Patients who received ivermectin required a lesser number of days (8.39 days ± 2.04) to become COVID negative than the patients who didn't receive ivermectin (20.38 days ± 6.32), (p < 0.001). Multinomial logistic regression showed that the patients who were given ivermectin for COVID 19 infection were four times more likely to be discharged home than stepping up to ICU. The ICU step-up rate in the ivermectin group was found to be 3.7% compared to 13.04% in the non-ivermectin group. No significant differences in mortality were found. Conclusion: Treatment with ivermectin in COVID-19 infection is associated with improved outcomes in terms of reduction in duration of illness as well as the progression of disease severity.
References
Behera, Patro, Singh, Chandanshive, Kumar et al., Role of ivermectin in the prevention of COVID-19 infection among healthcare workers in India: A matched case-control study,
doi:10.1101/2020.10.29.20222661Caly, Druce, Catton, Jans, Wagstaff, The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro, Antiviral Res,
doi:10.1042/bj20120150Chen, Klein, Garibaldi, Li, Wu et al., Aging in COVID-19: Vulnerability, immunity and intervention, Ageing Res Rev,
doi:10.1016/j.arr.2020.101205Cohen, Hall, John, Rapoport, The Early Natural History of SARS-CoV-2 Infection: Clinical Observations from an Urban, Ambulatory COVID
Dr, Ruth, Pfau, Hospital, None
Inciardi, Adamo, Lupi, Cani, Pasquale et al., Characteristics and outcomes of patients hospitalized for COVID-19 and cardiac disease in Northern Italy, Eur Heart J,
doi:10.1093/eurheartj/ehaa388Kumanan, Rajasooriyar, Guruparan, Sreeharan, The Impact of COVID-19 on the Delivery of Critical Health Care: Experience from a Non-High-Income Country, Asia Pac J Public Health,
doi:10.1177/1010539520963626Lespine, Alvinerie, Sutra, Pors, Chartier, Influence of the route of administration on efficacy and tissue distribution of ivermectin in goat, Vet Parasitol,
doi:10.1016/j.vetpar.2004.11.028Lippi, Wong, Henry, Hypertension in patients with coronavirus disease 2019 (COVID-19): A pooled analysis, Pol Arch Intern Med,
doi:10.20452/pamw.15272Lv, Liu, Wang, Dang, Qiu et al., Ivermectin inhibits DNA polymerase UL42 of pseudorabies virus entrance into the nucleus and proliferation of the virus in vitro and vivo, Antiviral Res,
doi:10.1016/j.antiviral.2018.09.010Mbbs, Dr, None
Mbbs, Dr, None
Mbbs, Dr, None
Mbbs, Dr, None
Mbbs, Medical Officer, Ruth, Pfau, Hospital, None
Mbbs, Officer, Ruth, Pfau, Hospital, None
Mbbs, Officer, Ruth, Pfau, Hospital, None
Mbbs, Officer, Ruth, Pfau, Hospital, None
Patt, Gordan, Diaz, Okon, Grady et al., Impact of COVID-19 on Cancer Care: How the pandemic is delaying cancer diagnosis and treatment for American seniors, JCO Clin Cancer Inform,
doi:10.1200/cci.20.00134Rajter, Sherman, Fatteh, Vogel, Sacks et al., Use of ivermectin is associated with lower mortality in hospitalized patients with coronavirus disease 2019: The Ivermectin in COVID Nineteen Study, Chest,
doi:10.1016/j.chest.2020.10.009Wagstaff, Sivakumaran, Heaton, Harrich, Jans, Ivermectin is a specific inhibitor of importin α/β-mediated nuclear import able to inhibit replication of HIV-1 and dengue virus, Biochem J,
doi:10.1042/bj20120150Wang, Lv, Wang, Qiu, Yang, Ivermectin treatment inhibits the replication of Porcine circovirus 2 (PCV2) in vitro and mitigates the impact of viral infection in piglets, Virus Res,
doi:10.1016/j.virusres.2019.01.010DOI record:
{
"DOI": "10.29309/tpmj/2022.29.09.6634",
"ISSN": [
"2071-7733",
"1024-8919"
],
"URL": "http://dx.doi.org/10.29309/TPMJ/2022.29.09.6634",
"abstract": "<jats:p>Objective: To investigate the efficacy of ivermectin in the treatment of mild, moderate, and severe COVID-19 infection. Study Design: Retrospective Cohort study. Setting: COVID-19 Treatment Centre, Dr. Ruth K M Pfau Civil Hospital Karachi. Period: July 2020 to December 2020. Material & Methods: Medical records of 423 patients during a selected duration of 6 months were reviewed. Patients were stratified into two groups based on whether or not they received a 6-day course of ivermectin in addition to the standard treatment for COVID-19. Primary outcome measures were rate of mortality, days from the start of treatment to negative SARS-CoV-2 PCR, and rate of step-up to the intensive care unit. Results: Patients who received ivermectin required a lesser number of days (8.39 days ± 2.04) to become COVID negative than the patients who didn't receive ivermectin (20.38 days ± 6.32), (p < 0.001). Multinomial logistic regression showed that the patients who were given ivermectin for COVID 19 infection were four times more likely to be discharged home than stepping up to ICU. The ICU step-up rate in the ivermectin group was found to be 3.7% compared to 13.04% in the non-ivermectin group. No significant differences in mortality were found. Conclusion: Treatment with ivermectin in COVID-19 infection is associated with improved outcomes in terms of reduction in duration of illness as well as the progression of disease severity.</jats:p>",
"author": [
{
"affiliation": [],
"family": "Tehseen Akhtar",
"sequence": "first"
},
{
"affiliation": [],
"family": "Amber Hanif",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pyar Ali",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Muhammad Hussain Haroon",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nimra Akram",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Muhammad Umar Raza",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Khalid Imran",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nimra Shakeel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ramsha Shakeel",
"sequence": "additional"
}
],
"container-title": "The Professional Medical Journal",
"container-title-short": "TPMJ",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
9,
2
]
],
"date-time": "2022-09-02T18:24:46Z",
"timestamp": 1662143086000
},
"deposited": {
"date-parts": [
[
2022,
9,
2
]
],
"date-time": "2022-09-02T18:24:46Z",
"timestamp": 1662143086000
},
"indexed": {
"date-parts": [
[
2022,
9,
2
]
],
"date-time": "2022-09-02T18:42:42Z",
"timestamp": 1662144162310
},
"is-referenced-by-count": 0,
"issue": "09",
"issued": {
"date-parts": [
[
2022,
9,
1
]
]
},
"journal-issue": {
"issue": "09",
"published-online": {
"date-parts": [
[
2022,
9,
1
]
]
}
},
"license": [
{
"URL": "https://creativecommons.org/licenses/by-nc/4.0",
"content-version": "unspecified",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
9,
1
]
],
"date-time": "2022-09-01T00:00:00Z",
"timestamp": 1661990400000
}
}
],
"link": [
{
"URL": "http://theprofesional.com/index.php/tpmj/article/download/6634/4970",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "http://theprofesional.com/index.php/tpmj/article/download/6634/4970",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "11864",
"original-title": [],
"page": "1384-1391",
"prefix": "10.29309",
"published": {
"date-parts": [
[
2022,
9,
1
]
]
},
"published-online": {
"date-parts": [
[
2022,
9,
1
]
]
},
"publisher": "Independent Medical Trust",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "http://theprofesional.com/index.php/tpmj/article/view/6634"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Pulmonary and Respiratory Medicine",
"Pediatrics, Perinatology and Child Health"
],
"subtitle": [],
"title": "Does ivermectin reduce COVID-19 mortality and progression of disease severity? – A retrospective study.",
"type": "journal-article",
"volume": "29"
}