Clinical Risk and Outpatient Therapy Utilization for COVID-19 in the Medicare Population
PhD; Andrew D Wilcock, PhD Stephen Kissler, MD Ateev Mehrotra, PT Brian E Mcgarry, PhD Benjamin D Sommers, PhD David C Grabowski, PhD Yonatan H Grad, MD Michael L Barnett
JAMA Health Forum, doi:10.1001/jamahealthforum.2023.5044
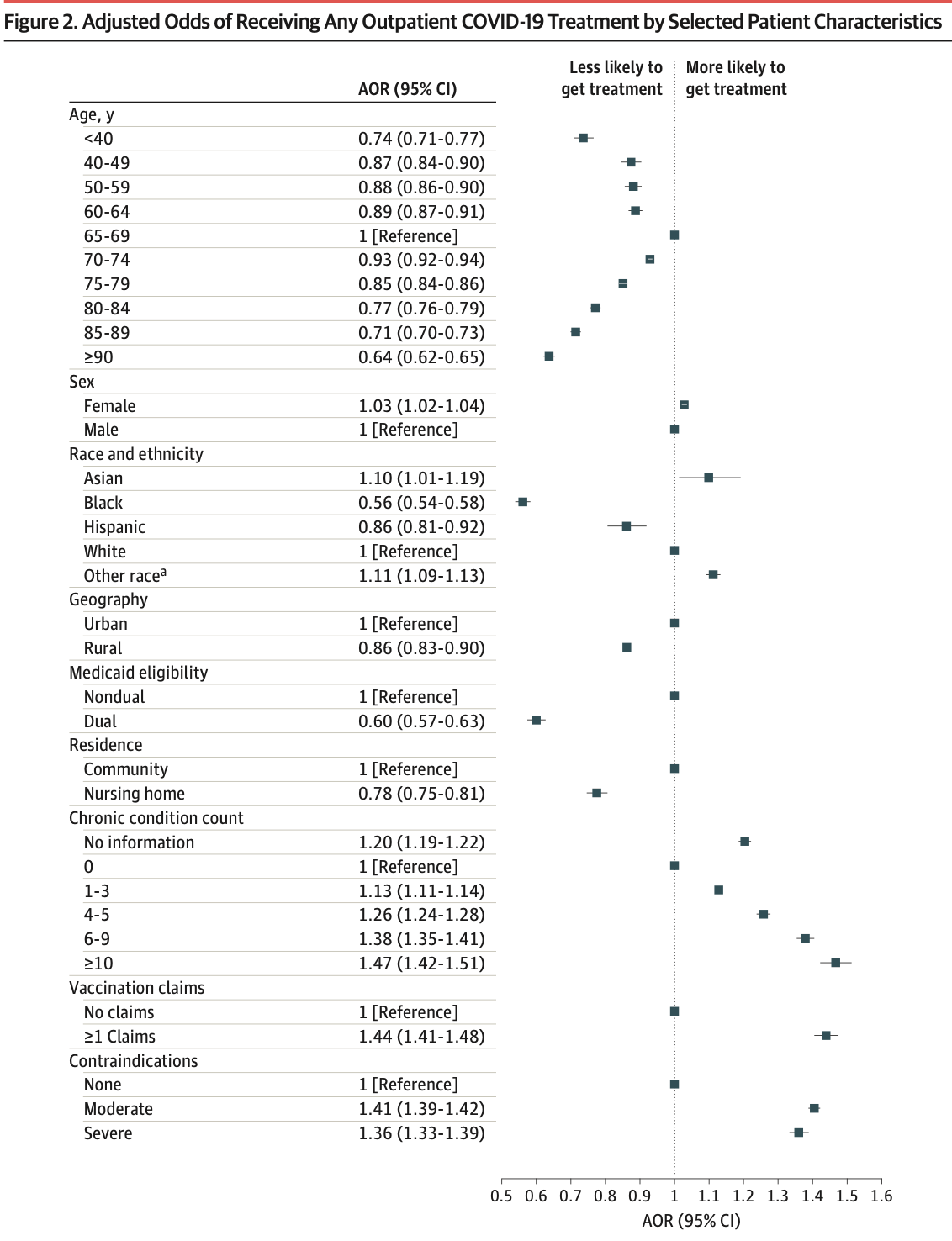
IMPORTANCE Multiple therapies are available for outpatient treatment of COVID-19 that are highly effective at preventing hospitalization and mortality. Although racial and socioeconomic disparities in use of these therapies have been documented, limited evidence exists on what factors explain differences in use and the potential public health relevance of these differences. OBJECTIVE To assess COVID-19 outpatient treatment utilization in the Medicare population and simulate the potential outcome of allocating treatment according to patient risk for severe COVID-19. DESIGN, SETTING, AND PARTICIPANTS This cross-sectional study included patients enrolled in Medicare in 2022 across the US, identified with 100% Medicare fee-for-service claims. MAIN OUTCOMES AND MEASURES The primary outcome was any COVID-19 outpatient therapy utilization. Secondary outcomes included COVID-19 testing, ambulatory visits, and hospitalization. Differences in outcomes were estimated based on patient demographics, treatment contraindications, and a composite risk score for mortality after COVID-19 based on demographics and comorbidities. A simulation of reallocating COVID-19 treatment, particularly with nirmatrelvir, to those at high risk of severe disease was performed, and the potential COVID-19 hospitalizations and mortality outcomes were assessed. RESULTS In 2022, 6.0% of 20 026 910 beneficiaries received outpatient COVID-19 treatment, 40.5% of which had no associated COVID-19 diagnosis within 10 days. Patients with higher risk for severe disease received less outpatient treatment, such as 6.4% of those aged 65 to 69 years compared with 4.9% of those 90 years and older (adjusted odds ratio [aOR], 0.64 [95% CI, 0.62-0.65]) and 6.4% of White patients compared with 3.0% of Black patients (aOR, 0.56 [95% CI, 0.54-0.58]). In the highest COVID-19 severity risk quintile, 2.6% were hospitalized for COVID-19 and 4.9% received outpatient treatment, compared with 0.2% and 7.5% in the lowest quintile. These patterns were similar among patients with a documented COVID-19 diagnosis, those with no claims for vaccination, and patients who are insured with Medicare Advantage. Differences were not explained by variable COVID-19 testing, ambulatory visits, or treatment contraindications. Reallocation of 2022 outpatient COVID-19 treatment, particularly with nirmatrelvir, based on risk for severe COVID-19 would have averted 16 503 COVID-19 deaths (16.3%) in the sample. CONCLUSION In this cross-sectional study, outpatient COVID-19 treatment was disproportionately accessed by beneficiaries at lower risk for severe infection, undermining its potential public health benefit. Undertreatment was not driven by lack of clinical access or treatment contraindications.
References
Abraham, Nohria, Neilan, Cardiovascular drug interactions with nirmatrelvir/ritonavir in patients with COVID-19: JACC review topic of the week, J Am Coll Cardiol,
doi:10.1016/j.jacc.2022.08.800Aggarwal, Molina, Beaty, Real-world use of nirmatrelvir-ritonavir in outpatients with COVID-19 during the era of omicron variants including BA.4 and BA.5 in Colorado, USA: a retrospective cohort study, Lancet Infect Dis,
doi:10.1016/S1473-3099(23)00011-7Arbel, Sagy, Hoshen, Nirmatrelvir use and severe Covid-19 outcomes during the omicron surge, N Engl J Med,
doi:10.1056/NEJMoa2204919Bajema, Berry, Streja, Effectiveness of COVID-19 treatment with nirmatrelvir-ritonavir or molnupiravir among US veterans: target trial emulation studies with one-month and six-month outcomes, Ann Intern Med,
doi:10.7326/M22-3565Barnett, Bitton, Souza, Landon, Trends in outpatient care for Medicare beneficiaries and implications for primary care, 2000 to 2019, Ann Intern Med,
doi:10.7326/M21-1523Beleche, Bush, Finegold, Understanding coverage considerations for COVID-19 vaccines and treatments
Berenbrok, Tang, Gabriel, Access to community pharmacies: a nationwide geographic information systems cross-sectional analysis, J Am Pharm Assoc,
doi:10.1016/j.japh.2022.07.003Bernal, Da Silva, Musungaie, Molnupiravir for oral treatment of Covid-19 in nonhospitalized patients, N Engl J Med,
doi:10.1056/NEJMoa2116044Boehmer, Koumans, Skillen, Racial and ethnic disparities in outpatient treatment of COVID-19-United States, January-July 2022, MMWR Morb Mortal Wkly Rep,
doi:10.15585/mmwr.mm7143a2Dryden-Peterson, Kim, Kim, Nirmatrelvir plus ritonavir for early COVID-19 in a large US health system: a population-based cohort study, Ann Intern Med,
doi:10.7326/M22-2141Gold, Kelleher, Magid, Dispensing of oral antiviral drugs for treatment of COVID-19 by zip codelevel social vulnerability-United States, December 23, 2021, MMWR Morb Mortal Wkly Rep,
doi:10.15585/mmwr.mm7125e1Hammond, Leister-Tebbe, Gardner, Oral nirmatrelvir for high-risk, nonhospitalized adults with Covid-19, N Engl J Med,
doi:10.1056/NEJMoa2118542Jarrín, Nyandege, Grafova, Dong, Lin, Validity of race and ethnicity codes in Medicare administrative data compared with gold-standard self-reported race collected during routine home health care visits, Med Care,
doi:10.1097/MLR.0000000000001216Luisi, Sullivan, Sanchez, Use of COVIDTests.gov at-home test kits among adults in a national household probability sample-United States, 2022, MMWR Morb Mortal Wkly Rep,
doi:10.15585/mmwr.mm7216a6Murphy, Samson, Sommers, COVID-19 antivirals utilization: geographic and demographic patterns of treatment in 2022
Najjar-Debbiny, Gronich, Weber, Effectiveness of Paxlovid in reducing severe coronavirus disease 2019 and mortality in high-risk patients, Clin Infect Dis,
doi:10.1093/cid/ciac443Patel, Mehrotra, Huskamp, Uscher-Pines, Ganguli et al., Variation in telemedicine use and outpatient care during the COVID-19 pandemic in the United States, Health Aff (Millwood),
doi:10.1377/hlthaff.2020.01786Silk, Scobie, Duck, COVID-19 surveillance after expiration of the public health emergency declaration-United States, May 11, 2023, MMWR Morb Mortal Wkly Rep,
doi:10.15585/mmwr.mm7219e1Sullivan, Perrine, Kelleher, Notes from the field: dispensing of oral antiviral drugs for treatment of COVID-19 by zip code-level social vulnerability-United States, December 23, 2021, MMWR Morb Mortal Wkly Rep,
doi:10.15585/mmwr.mm7143a3Tarabichi, Kaelber, Thornton, Early racial and ethnic disparities in the prescription of nirmatrelvir for COVID-19, J Gen Intern Med,
doi:10.1007/s11606-022-07844-3Wiltz, Feehan, Molinari, Racial and ethnic disparities in receipt of medications for treatment of COVID-19-United States, March 2020-August 2021, MMWR Morb Mortal Wkly Rep,
doi:10.15585/mmwr.mm7103e1{ 'indexed': {'date-parts': [[2024, 1, 27]], 'date-time': '2024-01-27T00:03:18Z', 'timestamp': 1706313798695},
'reference-count': 36,
'publisher': 'American Medical Association (AMA)',
'issue': '1',
'content-domain': {'domain': [], 'crossmark-restriction': False},
'abstract': '<jats:sec id="ab-aoi230094-4"><jats:title>Importance</jats:title><jats:p>Multiple therapies '
'are available for outpatient treatment of COVID-19 that are highly effective at preventing '
'hospitalization and mortality. Although racial and socioeconomic disparities in use of these '
'therapies have been documented, limited evidence exists on what factors explain differences '
'in use and the potential public health relevance of these '
'differences.</jats:p></jats:sec><jats:sec '
'id="ab-aoi230094-5"><jats:title>Objective</jats:title><jats:p>To assess COVID-19 outpatient '
'treatment utilization in the Medicare population and simulate the potential outcome of '
'allocating treatment according to patient risk for severe '
'COVID-19.</jats:p></jats:sec><jats:sec id="ab-aoi230094-6"><jats:title>Design, Setting, and '
'Participants</jats:title><jats:p>This cross-sectional study included patients enrolled in '
'Medicare in 2022 across the US, identified with 100% Medicare fee-for-service '
'claims.</jats:p></jats:sec><jats:sec id="ab-aoi230094-7"><jats:title>Main Outcomes and '
'Measures</jats:title><jats:p>The primary outcome was any COVID-19 outpatient therapy '
'utilization. Secondary outcomes included COVID-19 testing, ambulatory visits, and '
'hospitalization. Differences in outcomes were estimated based on patient demographics, '
'treatment contraindications, and a composite risk score for mortality after COVID-19 based on '
'demographics and comorbidities. A simulation of reallocating COVID-19 treatment, particularly '
'with nirmatrelvir, to those at high risk of severe disease was performed, and the potential '
'COVID-19 hospitalizations and mortality outcomes were assessed.</jats:p></jats:sec><jats:sec '
'id="ab-aoi230094-8"><jats:title>Results</jats:title><jats:p>In 2022, 6.0% of 20\u202f026\u202f'
'910 beneficiaries received outpatient COVID-19 treatment, 40.5% of which had no associated '
'COVID-19 diagnosis within 10 days. Patients with higher risk for severe disease received less '
'outpatient treatment, such as 6.4% of those aged 65 to 69 years compared with 4.9% of those '
'90 years and older (adjusted odds ratio [aOR], 0.64 [95% CI, 0.62-0.65]) and 6.4% of White '
'patients compared with 3.0% of Black patients (aOR, 0.56 [95% CI, 0.54-0.58]). In the highest '
'COVID-19 severity risk quintile, 2.6% were hospitalized for COVID-19 and 4.9% received '
'outpatient treatment, compared with 0.2% and 7.5% in the lowest quintile. These patterns were '
'similar among patients with a documented COVID-19 diagnosis, those with no claims for '
'vaccination, and patients who are insured with Medicare Advantage. Differences were not '
'explained by variable COVID-19 testing, ambulatory visits, or treatment contraindications. '
'Reallocation of 2022 outpatient COVID-19 treatment, particularly with nirmatrelvir, based on '
'risk for severe COVID-19 would have averted 16\u202f503 COVID-19 deaths (16.3%) in the '
'sample.</jats:p></jats:sec><jats:sec '
'id="ab-aoi230094-9"><jats:title>Conclusion</jats:title><jats:p>In this cross-sectional study, '
'outpatient COVID-19 treatment was disproportionately accessed by beneficiaries at lower risk '
'for severe infection, undermining its potential public health benefit. Undertreatment was not '
'driven by lack of clinical access or treatment contraindications.</jats:p></jats:sec>',
'DOI': '10.1001/jamahealthforum.2023.5044',
'type': 'journal-article',
'created': {'date-parts': [[2024, 1, 26]], 'date-time': '2024-01-26T16:30:54Z', 'timestamp': 1706286654000},
'page': 'e235044',
'source': 'Crossref',
'is-referenced-by-count': 0,
'title': 'Clinical Risk and Outpatient Therapy Utilization for COVID-19 in the Medicare Population',
'prefix': '10.1001',
'volume': '5',
'author': [ { 'given': 'Andrew D.',
'family': 'Wilcock',
'sequence': 'first',
'affiliation': [{'name': 'Harvard Medical School, Boston, Massachusetts'}]},
{ 'given': 'Stephen',
'family': 'Kissler',
'sequence': 'additional',
'affiliation': [ { 'name': 'Harvard T.H. Chan School of Public Health, Boston, '
'Massachusetts'}]},
{ 'given': 'Ateev',
'family': 'Mehrotra',
'sequence': 'additional',
'affiliation': [ {'name': 'Harvard Medical School, Boston, Massachusetts'},
{'name': 'Beth Israel Deaconess Medical Center, Boston, Massachusetts'}]},
{ 'given': 'Brian E.',
'family': 'McGarry',
'sequence': 'additional',
'affiliation': [{'name': 'University of Rochester Medical Center, Rochester, New York'}]},
{ 'given': 'Benjamin D.',
'family': 'Sommers',
'sequence': 'additional',
'affiliation': [ { 'name': 'Harvard T.H. Chan School of Public Health, Boston, '
'Massachusetts'}]},
{ 'given': 'David C.',
'family': 'Grabowski',
'sequence': 'additional',
'affiliation': [{'name': 'Harvard Medical School, Boston, Massachusetts'}]},
{ 'given': 'Yonatan H.',
'family': 'Grad',
'sequence': 'additional',
'affiliation': [ { 'name': 'Harvard T.H. Chan School of Public Health, Boston, '
'Massachusetts'}]},
{ 'given': 'Michael L.',
'family': 'Barnett',
'sequence': 'additional',
'affiliation': [ {'name': 'Harvard T.H. Chan School of Public Health, Boston, Massachusetts'},
{ 'name': 'Division of General Internal Medicine and Primary Care, '
'Department of Medicine, Brigham and Women’s Hospital, Boston, '
'Massachusetts'}]}],
'member': '10',
'published-online': {'date-parts': [[2024, 1, 26]]},
'reference': [ { 'issue': '15',
'key': 'aoi230094r2',
'doi-asserted-by': 'publisher',
'first-page': '1397',
'DOI': '10.1056/NEJMoa2118542',
'article-title': 'Oral nirmatrelvir for high-risk, nonhospitalized adults with Covid-19.',
'volume': '386',
'author': 'Hammond',
'year': '2022',
'journal-title': 'N Engl J Med'},
{ 'issue': '6',
'key': 'aoi230094r3',
'doi-asserted-by': 'publisher',
'first-page': '509',
'DOI': '10.1056/NEJMoa2116044',
'article-title': 'Molnupiravir for oral treatment of Covid-19 in nonhospitalized '
'patients.',
'volume': '386',
'author': 'Jayk Bernal',
'year': '2022',
'journal-title': 'N Engl J Med'},
{ 'issue': '3',
'key': 'aoi230094r4',
'doi-asserted-by': 'publisher',
'first-page': '96',
'DOI': '10.15585/mmwr.mm7103e1',
'article-title': 'Racial and ethnic disparities in receipt of medications for treatment '
'of COVID-19—United States, March 2020-August 2021.',
'volume': '71',
'author': 'Wiltz',
'year': '2022',
'journal-title': 'MMWR Morb Mortal Wkly Rep'},
{ 'issue': '10',
'key': 'aoi230094r5',
'doi-asserted-by': 'publisher',
'first-page': '980',
'DOI': '10.1001/jama.2022.1243',
'article-title': 'Anti-SARS-CoV-2 monoclonal antibody distribution to high-risk Medicare '
'beneficiaries, 2020-2021.',
'volume': '327',
'author': 'Behr',
'year': '2022',
'journal-title': 'JAMA'},
{ 'issue': '6',
'key': 'aoi230094r8',
'doi-asserted-by': 'publisher',
'first-page': '1816',
'DOI': '10.1016/j.japh.2022.07.003',
'article-title': 'Access to community pharmacies: a nationwide geographic information '
'systems cross-sectional analysis.',
'volume': '62',
'author': 'Berenbrok',
'year': '2022',
'journal-title': 'J Am Pharm Assoc (2003)'},
{ 'issue': '43',
'key': 'aoi230094r9',
'doi-asserted-by': 'publisher',
'first-page': '1359',
'DOI': '10.15585/mmwr.mm7143a2',
'article-title': 'Racial and ethnic disparities in outpatient treatment of '
'COVID-19—United States, January-July 2022.',
'volume': '71',
'author': 'Boehmer',
'year': '2022',
'journal-title': 'MMWR Morb Mortal Wkly Rep'},
{ 'issue': '5',
'key': 'aoi230094r12',
'doi-asserted-by': 'publisher',
'first-page': '1329',
'DOI': '10.1007/s11606-022-07844-3',
'article-title': 'Early racial and ethnic disparities in the prescription of nirmatrelvir '
'for COVID-19.',
'volume': '38',
'author': 'Tarabichi',
'year': '2023',
'journal-title': 'J Gen Intern Med'},
{ 'issue': '25',
'key': 'aoi230094r13',
'doi-asserted-by': 'publisher',
'first-page': '825',
'DOI': '10.15585/mmwr.mm7125e1',
'article-title': 'Dispensing of oral antiviral drugs for treatment of COVID-19 by zip '
'code—level social vulnerability—United States, December 23, 2021-May '
'21, 2022.',
'volume': '71',
'author': 'Gold',
'year': '2022',
'journal-title': 'MMWR Morb Mortal Wkly Rep'},
{ 'issue': '43',
'key': 'aoi230094r14',
'doi-asserted-by': 'publisher',
'first-page': '1384',
'DOI': '10.15585/mmwr.mm7143a3',
'article-title': 'Notes from the field: dispensing of oral antiviral drugs for treatment '
'of COVID-19 by zip code-level social vulnerability—United States, '
'December 23, 2021-August 28, 2022.',
'volume': '71',
'author': 'Sullivan',
'year': '2022',
'journal-title': 'MMWR Morb Mortal Wkly Rep'},
{ 'issue': '24',
'key': 'aoi230094r15',
'doi-asserted-by': 'publisher',
'first-page': '2466',
'DOI': '10.1001/jama.2020.8598',
'article-title': 'COVID-19 and racial/ethnic disparities.',
'volume': '323',
'author': 'Webb Hooper',
'year': '2020',
'journal-title': 'JAMA'},
{ 'issue': '16',
'key': 'aoi230094r16',
'doi-asserted-by': 'publisher',
'first-page': '445',
'DOI': '10.15585/mmwr.mm7216a6',
'article-title': 'Use of COVIDTests.gov at-home test kits among adults in a national '
'household probability sample—United States, 2022.',
'volume': '72',
'author': 'Luisi',
'year': '2023',
'journal-title': 'MMWR Morb Mortal Wkly Rep'},
{ 'issue': '19',
'key': 'aoi230094r18',
'doi-asserted-by': 'publisher',
'first-page': '523',
'DOI': '10.15585/mmwr.mm7219e1',
'article-title': 'COVID-19 surveillance after expiration of the public health emergency '
'declaration—United States, May 11, 2023.',
'volume': '72',
'author': 'Silk',
'year': '2023',
'journal-title': 'MMWR Morb Mortal Wkly Rep'},
{ 'issue': '1',
'key': 'aoi230094r22',
'doi-asserted-by': 'publisher',
'first-page': 'e1',
'DOI': '10.1097/MLR.0000000000001216',
'article-title': 'Validity of race and ethnicity codes in Medicare administrative data '
'compared with gold-standard self-reported race collected during routine '
'home health care visits.',
'volume': '58',
'author': 'Jarrín',
'year': '2020',
'journal-title': 'Med Care'},
{ 'issue': '1p1',
'key': 'aoi230094r23',
'doi-asserted-by': 'publisher',
'first-page': '120',
'DOI': '10.1111/hesr.2010.46.issue-1p1',
'article-title': "The residential history file: studying nursing home residents' "
'long-term care histories.',
'volume': '46',
'author': 'Intrator',
'year': '2011',
'journal-title': 'Health Serv Res'},
{ 'issue': '2',
'key': 'aoi230094r24',
'doi-asserted-by': 'publisher',
'first-page': '349',
'DOI': '10.1377/hlthaff.2020.01786',
'article-title': 'Variation in telemedicine use and outpatient care during the COVID-19 '
'pandemic in the United States.',
'volume': '40',
'author': 'Patel',
'year': '2021',
'journal-title': 'Health Aff (Millwood)'},
{ 'issue': '3',
'key': 'aoi230094r25',
'doi-asserted-by': 'publisher',
'first-page': '388',
'DOI': '10.1001/jamainternmed.2020.5928',
'article-title': 'Trends in outpatient care delivery and telemedicine during the COVID-19 '
'pandemic in the US.',
'volume': '181',
'author': 'Patel',
'year': '2021',
'journal-title': 'JAMA Intern Med'},
{ 'issue': '5',
'key': 'aoi230094r26',
'doi-asserted-by': 'publisher',
'DOI': '10.1001/jamanetworkopen.2021.10330',
'article-title': 'Community factors associated with telemedicine use during the COVID-19 '
'pandemic.',
'volume': '4',
'author': 'Patel',
'year': '2021',
'journal-title': 'JAMA Netw Open'},
{ 'issue': '12',
'key': 'aoi230094r27',
'doi-asserted-by': 'publisher',
'first-page': '1658',
'DOI': '10.7326/M21-1523',
'article-title': 'Trends in outpatient care for Medicare beneficiaries and implications '
'for primary care, 2000 to 2019.',
'volume': '174',
'author': 'Barnett',
'year': '2021',
'journal-title': 'Ann Intern Med'},
{ 'issue': '20',
'key': 'aoi230094r28',
'doi-asserted-by': 'publisher',
'first-page': '1912',
'DOI': '10.1016/j.jacc.2022.08.800',
'article-title': 'Cardiovascular drug interactions with nirmatrelvir/ritonavir in '
'patients with COVID-19: JACC review topic of the week.',
'volume': '80',
'author': 'Abraham',
'year': '2022',
'journal-title': 'J Am Coll Cardiol'},
{ 'issue': '6',
'key': 'aoi230094r30',
'doi-asserted-by': 'publisher',
'first-page': '807',
'DOI': '10.7326/M22-3565',
'article-title': 'Effectiveness of COVID-19 treatment with nirmatrelvir-ritonavir or '
'molnupiravir among US veterans: target trial emulation studies with '
'one-month and six-month outcomes.',
'volume': '176',
'author': 'Bajema',
'year': '2023',
'journal-title': 'Ann Intern Med'},
{ 'issue': '1',
'key': 'aoi230094r31',
'doi-asserted-by': 'publisher',
'first-page': '77',
'DOI': '10.7326/M22-2141',
'article-title': 'Nirmatrelvir plus ritonavir for early COVID-19 in a large US health '
'system: a population-based cohort study.',
'volume': '176',
'author': 'Dryden-Peterson',
'year': '2023',
'journal-title': 'Ann Intern Med'},
{ 'issue': '9',
'key': 'aoi230094r32',
'doi-asserted-by': 'publisher',
'first-page': '790',
'DOI': '10.1056/NEJMoa2204919',
'article-title': 'Nirmatrelvir use and severe Covid-19 outcomes during the omicron surge.',
'volume': '387',
'author': 'Arbel',
'year': '2022',
'journal-title': 'N Engl J Med'},
{ 'issue': '6',
'key': 'aoi230094r33',
'doi-asserted-by': 'publisher',
'first-page': '696',
'DOI': '10.1016/S1473-3099(23)00011-7',
'article-title': 'Real-world use of nirmatrelvir-ritonavir in outpatients with COVID-19 '
'during the era of omicron variants including BA.4 and BA.5 in Colorado, '
'USA: a retrospective cohort study.',
'volume': '23',
'author': 'Aggarwal',
'year': '2023',
'journal-title': 'Lancet Infect Dis'},
{ 'issue': '3',
'key': 'aoi230094r34',
'doi-asserted-by': 'publisher',
'first-page': 'e342',
'DOI': '10.1093/cid/ciac443',
'article-title': 'Effectiveness of Paxlovid in reducing severe coronavirus disease 2019 '
'and mortality in high-risk patients.',
'volume': '76',
'author': 'Najjar-Debbiny',
'year': '2023',
'journal-title': 'Clin Infect Dis'},
{ 'key': 'aoi230094r1',
'unstructured': 'National Institutes of Health. Nonhospitalized adults—therapeutic '
'management. COVID-19 Treatment Guidelines. Accessed May 1, 2023. '
'https://www.covid19treatmentguidelines.nih.gov/management/clinical-management-of-adults/nonhospitalized-adults--therapeutic-management/'},
{ 'key': 'aoi230094r6',
'unstructured': 'Coronavirus (COVID-19) update: FDA authorizes first oral antiviral for '
'treatment of COVID-19. US Food and Drug Administration. 2021. May 1, '
'2023. '
'https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-authorizes-first-oral-antiviral-treatment-covid-19'},
{ 'key': 'aoi230094r7',
'unstructured': 'Coronavirus (COVID-19) update: FDA authorizes additional oral antiviral '
'for treatment of COVID-19 in certain adults. FDA. 2021 May 1, 2023. '
'https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-authorizes-additional-oral-antiviral-treatment-covid-19-certain'},
{ 'key': 'aoi230094r10',
'unstructured': 'COVID-19 antivirals utilization: geographic and demographic patterns of '
'treatment in 2022. ASPE. Accessed April 10, 2023. '
'https://aspe.hhs.gov/reports/covid-19-antivirals-utilization'},
{ 'key': 'aoi230094r11',
'unstructured': 'FDA takes actions to expand use of treatment for outpatients with '
'mild-to-moderate COVID-19. US Food and Drug Administration. 2022. '
'Accessed November 14, 2023. '
'https://www.fda.gov/news-events/press-announcements/fda-takes-actions-expand-use-treatment-outpatients-mild-moderate-covid-19'},
{ 'key': 'aoi230094r17',
'unstructured': 'COVID data tracker. Centers for Disease Control and Prevention. 2020. '
'Accessed March 8, 2023. https://covid.cdc.gov/covid-data-tracker'},
{ 'key': 'aoi230094r19',
'unstructured': 'COVID-19 information and resources. Harvard Pilgrim Health Care. '
'Accessed April 10, 2023. '
'https://www.point32health.org/provider/news-center/coronavirus-covid-19-updates-for-providers/'},
{ 'key': 'aoi230094r20',
'unstructured': 'UW RHRC Rural Urban Commuting Area Codes. UW RUCA. Accessed May 5, 2023. '
'https://depts.washington.edu/uwruca/'},
{ 'key': 'aoi230094r21',
'unstructured': 'Chronic conditions. Chronic Conditions Data Warehouse. Accessed December '
'11, 2022. '
'https://www2.ccwdata.org/web/guest/condition-categories-chronic'},
{ 'key': 'aoi230094r29',
'unstructured': 'Paxlovid drug-drug interactions. COVID-19 Treatment Guidelines. Accessed '
'April 10, 2023. Updated November 2, 2023. '
'https://www.covid19treatmentguidelines.nih.gov/therapies/antivirals-including-antibody-products/ritonavir-boosted-nirmatrelvir--paxlovid-/paxlovid-drug-drug-interactions/'},
{ 'key': 'aoi230094r35',
'unstructured': 'Murphy? SJ, Samson? LW, Sommers? BD. COVID-19 antivirals utilization: '
'geographic and demographic patterns of treatment in 2022. Accessed '
'December 13, 2023. '
'https://aspe.hhs.gov/reports/covid-19-antivirals-utilization'},
{ 'key': 'aoi230094r36',
'unstructured': 'Beleche? T, Bush? L, Finegold? K, Understanding coverage considerations '
'for COVID-19 vaccines and treatments. 2022. '
'https://aspe.hhs.gov/reports/covid-19-vaccines-treatments'}],
'container-title': 'JAMA Health Forum',
'original-title': [],
'language': 'en',
'link': [ { 'URL': 'https://jamanetwork.com/journals/jama-health-forum/articlepdf/2814359/wilcock_2024_oi_230094_1706287370.16897.pdf',
'content-type': 'unspecified',
'content-version': 'vor',
'intended-application': 'similarity-checking'}],
'deposited': { 'date-parts': [[2024, 1, 26]],
'date-time': '2024-01-26T17:00:41Z',
'timestamp': 1706288441000},
'score': 1,
'resource': {'primary': {'URL': 'https://jamanetwork.com/journals/jama-health-forum/fullarticle/2814359'}},
'subtitle': [],
'short-title': [],
'issued': {'date-parts': [[2024, 1, 26]]},
'references-count': 36,
'journal-issue': {'issue': '1', 'published-print': {'date-parts': [[2024, 1, 5]]}},
'URL': 'http://dx.doi.org/10.1001/jamahealthforum.2023.5044',
'relation': {},
'ISSN': ['2689-0186'],
'subject': ['Public Health, Environmental and Occupational Health', 'Health Policy'],
'container-title-short': 'JAMA Health Forum',
'published': {'date-parts': [[2024, 1, 26]]}}